In order to carry out clinical trials of regenerative medical products safely and promptly, it is necessary to have expert knowledge and familiarity with operation processes at medical institutions, while it is also required to accumulate experiences together with the sponsor and medical institutions.
When intellim began working on clinical trials of viral vector products as a CRO in 2014, sponsors were developing development strategies of regenerative medicine products through trial and error. However, it can be said that the environment for the implementation of regenerative medicine trials including investigator-initiated clinical trials and clinical researches at intellim Group has been well established.
In this article, we will explain the process until intellim established the environment to conduct the development of the regenerative medicine field and the training system for CRAs.
Start with enhancing support for the revision of SOPs
In a broad sense, clinical researches in the field of regenerative medicine must be conducted in accordance with three major laws and regulations (GCP, the Act on Securement of Safety of Regenerative Medicine, the Clinical Trials Act and the Cartagena Act in the case of viral vector products).
However, when intellim first started full-scale development of regenerative medicine, we found some issues. The issues are that only very few medical institutions had SOPs that met the standards of GCP clinical trials for regenerative medicine or medical institutions did not have SOPs although they had medical specialists.
Therefore, intellim started with enhancing support for medical institutions to revise their SOPs.
By simultaneously enhancing support for the establishment of clinical trial implementation systems, we have been able to smoothly receive cooperation in revising SOPs from medical institutions and other facilities.
For clinical trial sponsors who do not have experiences in the development of regenerative medicine products, we strive to alleviate the concerns of medical institution staff by providing them with information on different things from regulations in pharmaceutical trials and relevant points to note and concerns that medical institutions may have.
In 2017, a department specializing in regenerative medicine was established at intellim. The department will accumulate experiences through our contracted projects. "Regenerative Medicine Specialist Certification System" was also established to prepare a system to respond to the increasing number of clinical trials related to regenerative medicine products in the future.
“Regenerative Medicine Specialist" starting from the basics
As mentioned above, there are many differences and points to note in regenerative medicine clinical trials from the regulations of general drug clinical trials.
In intellim's "Regenerative Medicine Specialist Training", our CRAs learn the basic knowledge and terminology that are important for understanding regenerative medicine, such as cells and viral vectors.
Since regenerative medicine laws include a lot of unique terminologies, CRAs must have basic knowledge and understanding of the terminologies beforehand in order to properly understand the regulations.
Furthermore, in order to understand the basics of regenerative medicine, knowledge of "basic biology" is also essential. Therefore, intellim has also prepared "Basic Biology Training" for personnel who are not confident about basic biology or whose knowledge is deemed insufficient. In addition to the rigorous certification system, we have established a system that allows any personnel with interest and motivation in regenerative medicine to challenge the certification as a regenerative medicine specialist.
Such training sessions starting from the basics helped us establish the cycle where we can produce an effect in supporting the revision of SOPs and the establishment of clinical trial implementation systems and then it leads to the experience of creating even better human resources.
“Regenerative Medicine Specialist Training" is divided into several sessions including basic, legal, and quality/safety sessions. The legal session begins with a general overview of the laws that must be followed when implementing clinical trials of regenerative medicine products. In the quality and safety session, we unravel the contents and items of non-clinical studies that must be performed prior the First In Human study, as well as the contents and items of quality inspections conducted in the process of product manufacturing based on the guidelines.

Until provision of "Regenerative Medicine Specialist Certification System" to clients
"Regenerative Medicine Specialist Certification System," which was created in the above way, has also led to a higher evaluation of our CRAs by clients. As the result, more and more investigators and clients are interested in our training system, and are asking us if we can provide them with training secessions.
Therefore, we decided to offer "Regenerative Medicine Specialist Certification System" which was originally designed for our internal training program to external customers.
In fact, since regenerative medicine is still a new field and there is no accumulated data of regenerative medicine, it is difficult for many companies to give training secessions about regenerative medicine to their employees on their own. Therefore, intellim's experience and knowledge have been highly valued by our clients.
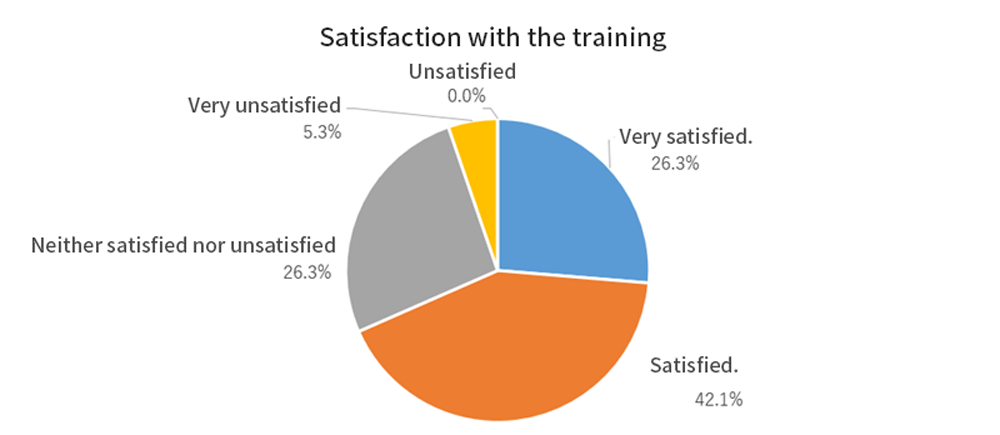
At intellim, we have opportunities to hear about the words and expressions of patients who have received the most advanced medical care from their investigators. We have heard more and more stories like that patients received very satisfying results from radically curing diseases which had only few treatment options and no fundamental solution.
Hearing such comments is a great pleasure and a rewarding experience for CRAs.
If you are interested in our regenerative medicine specialist training, please contact us via the <link to contact form>(This will open in a new window.).